
NEWS
最新消息
Prodrug, Metabolite and Intermediate Patent Infringement
- 分类:最新消息
- 作者:华讯知识产权
- 来源:
- 发布时间:2019-08-06 09:15
- 访问量:
【概要描述】ProdrugAprodrugisdefinedasapharmacologicallyinactiveorlightlyactivecompoundthatisthechemicallymodifiedformofapharmacologicallyactivedrugtowhichitisconverted(asbyenzymaticornonenzymaticaction)inthebody
Prodrug, Metabolite and Intermediate Patent Infringement
【概要描述】ProdrugAprodrugisdefinedasapharmacologicallyinactiveorlightlyactivecompoundthatisthechemicallymodifiedformofapharmacologicallyactivedrugtowhichitisconverted(asbyenzymaticornonenzymaticaction)inthebody
- 分类:最新消息
- 作者:华讯知识产权
- 来源:
- 发布时间:2019-08-06 09:15
- 访问量:
Prodrug
A prodrug is defined as a pharmacologically inactive or lightly active compound that is the chemically modified form of a pharmacologically active drug to which it is converted (as by enzymatic or nonenzymatic action) in the body.
Subject to the applicable provisions of the Patent Law, the compound used in the drug patent of invention means a single substance with certain chemical structure or physical or chemical property, including the prodrug.

Prodrug Patent Infringement
The prodrug, after being taken by a patient, plays its curative effects after being converted to the parent drug after such processes as in vivo absorption, metabolization, distribution, etc. So, whether does generic drug enterprises’ preparation of prodrugs infringe upon the patent rights of the core patent of the compounds of brand drug enterprises, thus becoming a method of avoiding active compounds?
The traditional patent infringement is judged based on the universal coverage principle that restricts the extent of protection of the patent rights based on the claims as the benchmark. Only the technical characteristics of the products or methods that are accused of infringement correspond to and are identical to all of the technical characteristics as recorded by the claims, or the technical characteristics of the accused subject are included in all of the technical characteristics as recorded in the claims and added some other technical characteristics, it constitutes the infringement. Based on the above, literally, the prodrug is not falling within the extent of protection of the core patent of compounds due to its different molecular formula.
In addition to the universal coverage principle, the infringement judgment also relates to the doctrine of equivalents. The so called “doctrine of equivalents” means that although the technical characteristics of the accused subject are different from all of necessary technical characteristics as recorded by the claims, such difference is not substantive, and the former is just the basically same means as that of the latter, and realizes the basically same functions and effects, and the characteristics are those that the ordinary technicians in this field can image without any creative labor, i.e. equivalent characteristics, it still can be judged as such actions of infringement nature. However, since the prodrug can obviously enhance such nature of the compound as pharmacokinetics, etc., the doctrine of equivalents is not applicable to the prodrug possibly due to its “unexpected technical effects”.
However, looking from the effects the prodrug plays finally, no matter which form of prodrug the generic drug enterprises provide, the molecules that play the active role in the body of a patient are those chemical molecules protected by the core patent of the compound. If the prodrug is not involved in the infringement, it is hard to avoid damages to the innovation subject. This is inconsistent with the original spirit of the patent law that is set up to encourage invention and creation.
In this respect, the “contributory infringement” regime solves to some extent the above issue.
Up to date, the Patent Law sets forth no provisions about the contributory infringement. The Article 130 of the General Principles of the Civil Law of the People’s Republic of China: “if two or more persons jointly infringe upon another person's rights and cause him damage, they shall bear joint and several liabilities”, the Article 148 of the Opinions on Carrying out and Implementing Numerous Issues of the General Principles of the Civil Law of the People’s Republic of China: “one who abets or assists another person in committing a tort will be the joint tortfeasor and shall bear the joint and several civil liabilities,” and the Article 9 of the Tort Law of the People’s Republic of China: “one who abets or assists another person in committing a tort shall be liable jointly and severally with the tortfeasor”, will be regarded as the legal basis of judging the establishment of the contributory infringement and investigating into the tort liabilities.

Infringement Case
This is the case of Beecham against Bristol as judged by the British House of Lords in 1977. Since Hetacillin is converted to Ampicillin in the body, the court judged that the prodrug – Hetacillin infringed upon the patents of Ampicillin, and determined that the sales of Hetacillin infringed upon others’ patent rights, on the ground that when the prodrug contacts the water in the esophagus, and after chemical reaction, becomes the active Ampicillin. The court held that Hetacillin had no treatment value or other added values. Although the patent stated in the claims is structurally different from the accused product, materially, the substance that plays the active role still utilizes the patent right of Ampicillin.
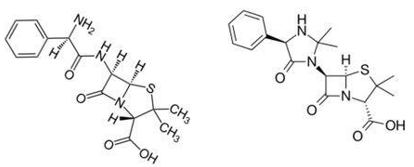
Fig. 1. Ampicillin (Left) and Hetacillin (Right) the latter is formed by condensing the former and acetone.
Now in China, there has been no infringement lawsuit case about the in vivo metabolic compounds. There is a similar case of the patent infringement dispute between Chongqing Xinyuanxing Pharmaceutical Co., Ltd. and Novartis on the subject of whether the intermediate not falling within the extent of protection of the sales rights claim infringes upon the patent rights.
Intermediate Patent Infringement Case
Novartis has the patent of the invention – “pyrimidine derivatives and their preparation methods and purposes”, i.e. the famous Imatinib (Glivec). In 2007, the evidence collectors from Novartis bought the samples of hydride (amino), piperidin ylbenzoicacid and Glivec. In the trial of first instance, Xinyuanxing admitted that the structural formulas of hydride (amino), piperidin ylbenzoicacid and Glivec provided by it were consistent with those as claimed in the webpage and publicity materials. The court of first instance held that the Imatinib and Glivec products manufactured by Xinyuanxing without authorization fell within the extent of protection of the patent rights, they constituted an infringement.
About the nature of the acts of Xinyuanxing manufacturing, selling and promising to sell the intermediate hydride (amino), piperidin ylbenzoicacid and intermediate nitro without authorization, since such three intermediates have no purposes other than preparation of Imatinib and Glivec, the sales of such three intermediates will consequentially cause the buyers to manufacture the Imatinib and Glivec products infringing upon the patent rights of the plaintiff by using them, and Xinyuanxing clearly stated at the website that the said intermediates were those used to manufacture Imatinib and Glivec, so Xinyuanxing knew the said consequences caused by its acts. Based on the above, although the said intermediates are not directly falling within the extent of protection of the patent of the plaintiff, it still constitutes the contributory infringement.
Xinyuanxing disputed the first trial decision, and appealed to Chongqing Higher People's Court, claiming that the intermediates had other purposes, so it didn’t constitute an infringement, and submitted an approval specification of the invention patent application for “method of preparation of benzothiazine dioxide derivatives”. The Chongqing Higher People's Court finally recognized that piperidin ylbenzoicacid had other purposes, but even if so, Xinyuanxing cleared at the website or in the written publicity materials and actual sales that piperidin ylbenzoicacid was sold as the intermediate of Glivec. Therefore, Xinyuanxing’s sales of and promise to sell piperidin ylbenzoicacid were to induce the occurrence of direct infringement. It constitutes the contributory infringement upon the patent concerned.

Conclusions
As analyzed above, if the drug manufacturers developed active prodrugs and sold them to patients, after being taken by patients, they are unavoidably converted to the parent drugs with the treatment effect, and the parent drugs are falling within the extent of protection of the core patent of compound. Then the sole purpose of preparing and selling the prodrug on the part of the drug manufacturer is to generate the patent drug, under which case, there is a large risk of patent infringement, except that the drug manufacturer can make sufficient defense in respect of existing technology, or has sufficient evidences and reasons to declare that the patent of the parent drug is void; otherwise, it is unlikely to make a successful defense.


美国联邦巡回上诉法院表示娇生公司 全球畅销精神分裂症药物的专利可能无效

注射用醋酸地加瑞克专利无效口审将于本月底进行

外观专利如何进行海外布局?










